Calcium Carbonate contains the mineral calcium carbonate and operates in over-the-counter (OTC) functions within pharmaceuticals and nutraceuticals. Classified as an active pharmaceutical Ingredient (API) under pharmaceutical actives and precursors within pharmaceuticals and nutraceuticals technologies, it’s utilized in various pharmaceutical formulations for its calcium-based properties.
Calcium Carbonate
471-34-1
AICS (Australia), DSL (Canada), ECL (Korea), EINECS (EU), ENCS (Japan), IECSC (China), NDSL (Canada), NZIoC (New Zealand), PICCS (Phillipines), TCSI (Taiwan), TSCA (USA)
British Pharmacopeia (BP), California Proposition 65, European Pharmacopeia (Ph. Eur.), FCC Compliant, FDA 29 CFR 1910.1200, FSSAI, Generally Recognized As Safe (GRAS), United States Pharmacopeia (USP)
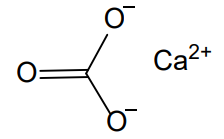
CaCO3
Enhanced TDS
Identification & Functionality
Chemical Family ;
Calcium Salts, Carbonates
Chemical Name :
Calcium Carbonate
Ingredient Name :
Calcium Carbonate
Mineral Type :
Calcium
Pharma & Nutraceuticals Functions :
OTC
Molecular formula :
CaCO₃
Technologies :
Food Ingredients, Pharmaceuticals & Nutraceuticals
Product Families :
Pharmaceuticals & Nutraceuticals — Nutraceuticals & Supplements
Minerals
Food Ingredients — Nutrition & Fortification
Minerals Pharmaceuticals & Nutraceuticals — Pharmaceutical Actives & Precursors Active Pharmaceutical Ingredients (APIs)
Definition :
Calcium Carbonate, dried at 200°C for 4 hours, contains calcium equivalent to not less than 98.0% and not more than 100.5% of calcium carbonate (CaCO₃).
Applications & Uses
Markets :
Food & Nutrition,
Healthcare & Pharma
Applications :
Healthcare & Pharma — Drug Delivery & Care Solutions
Other Drug & Care Solutions
Food & Nutrition — Nutrition & Well Being Nutraceuticals & Supplements
Properties
Physical Form :
Powder
Appearance :
White gran. powder
Regulatory & Compliance
Certifications & Compliance :
British Pharmacopeia (BP), California Proposition 65, European Pharmacopeia (Ph. Eur.), FCC Compliant, FDA 29 CFR 1910.1200, FSSAI, Generally Recognized As Safe (GRAS), United States Pharmacopeia (USP)
Chemical Inventories :
AICS (Australia), DSL(Canada), ECL (Korea), EINECS (EU), NCS (Japan), IECSC (China), NDSL (Canada), NZIoC (New Zealand), PICCS (Phillipines), TCSI (Taiwan), TSCA (USA)
FDA Disclaimer :
These statements have not been evaluated by the Food and Drug Administration. This product is not intended to diagnose, treat, cure, or prevent any disease.
USP Reference Standards :
USP Sodium Fluoride RS
Storage & Handling
Storage Information :
Preserve in well-closed containers.