Lactobacillus plantarum LPL28 is a product whose strain origin is isolated from miso with no microbial domestication or mutation process. Applications for this product include oral care, pathogen inhibition, vaginal health, and GABA production. The production process involves fermentation, centrifuging, freeze-drying, and packaging.
Lactobacillus Plantarum
Biodegradable, Halal, Kosher, Non-Hazardous, Non-Toxic
Probiotic
EFSA (European Food Safety Authority) Certified, FDA Compliant
Anti-Inflammatory, Improved Stability
| Value | Units | Test Method / Conditions | |
| Yeast & Molds Count | max. 100 | cfu/g | Taiwan Food and Drug Administration ( TFDA ) method |
| Staphylococcus Aureus | Negative | cfu/g | Taiwan Food and Drug Administration ( TFDA ) method |
| Salmonella | Negative | cfu/g | Taiwan Food and Drug Administration ( TFDA ) method |
| Listeria Monocytogenes | Negative | cfu/g | Taiwan Food and Drug Administration ( TFDA ) method |
| Eschericha Coli | Negative | cfu/g | Taiwan Food and Drug Administration ( TFDA ) method |
| Coliforms | Negative | cfu/g | Taiwan Food and Drug Administration ( TFDA ) method |
| Value | Units | Test Method / Conditions | |
| Water Activity | max. 0.25 | aw | Taiwan Food and Drug Administration ( TFDA ) method |
| Viable Cell Counts | max. 4.0 × 10¹¹ | cfu/g | Taiwan Food and Drug Administration ( TFDA ) method |
| Moisture Content | max. 7 | % | Taiwan Food and Drug Administration ( TFDA ) method |
| O. | Composition | Content (%) |
| 1 | Lactobacillus plantarum LPL28 | 100% |
| Total | 100% |
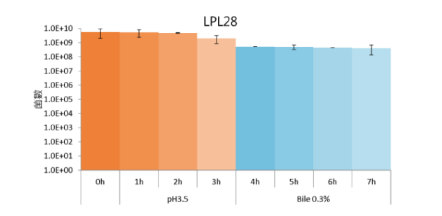
High Gastric Acid and Bile Tolerance:
LPL28 remained high bioavailability in the simulated environment with Gastric Acid and Bile for 7 hours
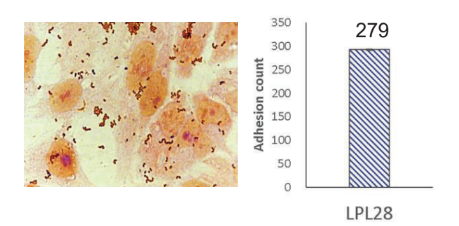
Strong Colonization on Caco-2 Intestine Cells:
LPL28 highly adheres to Caco-2 intestine cells
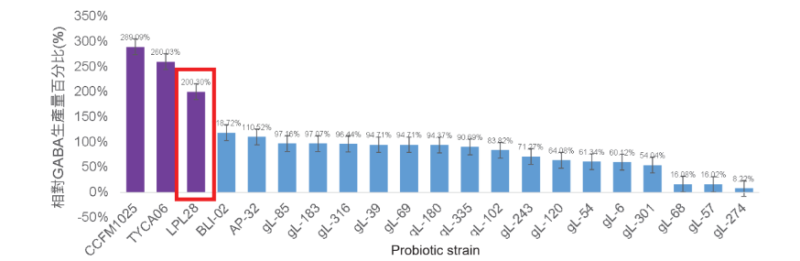
High GABA-producing probiotic strain:
1kg a bag with nitrogen filling and vacuum packing.
24 months
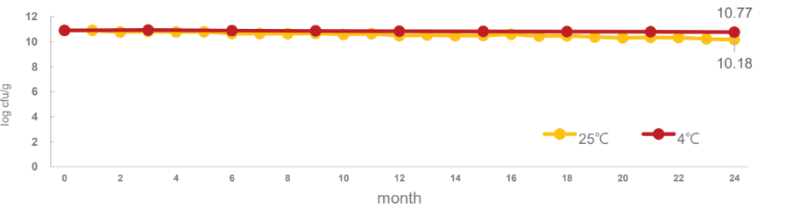
LPL28 remains stable at 4°C and 25°C for 24 months:
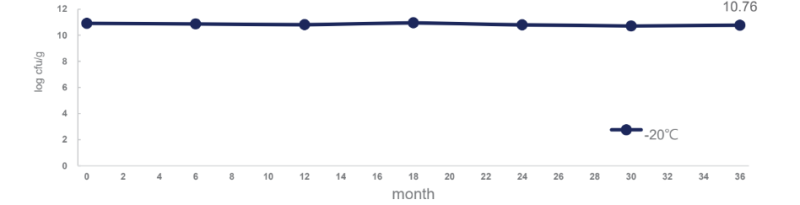
LPL28 remains stable at -20°C for 36 months: