Tetracaine operates in over-the-counter (OTC) capacities within pharmaceuticals and nutraceuticals. Composed of tetracaine, it falls under active pharmaceutical ingredients (APIs) within pharmaceutical actives and precursors. Pharm-Rx Tetracaine is commonly used in various pharmaceutical formulations for its anesthetic properties.
Amethocaine
94-24-6
AICS (Australia), EINECS (EU), TCSI (Taiwan)
California Proposition 65, FDA 29 CFR 1910.1200
Enhanced TDS
Identification & Functionality
Chemical Name :
Amethocaine
Pharma & Nutraceuticals Functions :
OTC
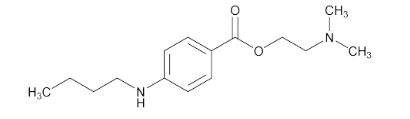
Molecular formula :
C₁₅H₂₄N₂O₂
Technologies :
Pharmaceuticals & Nutraceuticals
Product Families :
Pharmaceuticals & Nutraceuticals — Pharmaceutical Actives & Precursors
Active Pharmaceutical Ingredients (APIs)
Definition :
Tetracaine contains not less than 98.0% and not more than 102.0% of tetracaine (C₁₅H₂₄N₂O₂), calculated on the dried basis.
Applications & Uses
Markets :
Healthcare & Pharma
Applications :
Healthcare & Pharma — Drug Delivery & Care Solutions Other Drug & Care Solutions
Properties
Physical Form :
Powder
Soluble In :
Benzene Soluble, Chloroform Soluble, Ethanol Soluble, Ether Soluble, Trichloromethane Soluble